Lithium-ion batteries have grown in popularity since their creation in the 1970s. With several important advantages, such as being lightweight and able to hold a charge during long storage periods, lithium-based batteries are some of the most popular on the market.

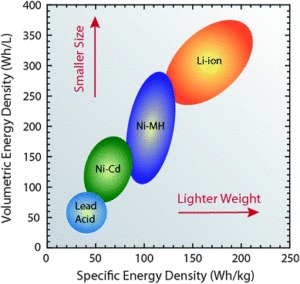
Lithium battery, lithium-polymer batteries, and lithium-ion batteries have emerged as popular energy sources in recent times. A regular lithium-ion battery can hold 150 watt-hours of electricity in a 1 kg battery, which is much higher compared to a lead acid battery with a storage capacity of 25 watt-hours per kg and NiMH battery with a storage capacity of 100 watt-hours/kg. With their many advantages, these batteries are now widely used in consumer electronics and battery-operated vehicles.
Lithium batteries are more popular today than ever before. You’ll find them in your cell phone, laptop computer, cordless power tools, and even electric vehicles. However, just because all of these electronics use lithium batteries doesn’t mean they use the same type of lithium batteries. We’ll take a closer look at the six main types of lithium batteries pros and cons, as well as the best applications for each.
What Is A Lithium Battery?
Lithium battery rely on lithium ions to store energy by creating an electrical potential difference between the negative and positive poles of the battery. An insulating layer called a “separator” divides the two sides of the battery and blocks the electrons while still allowing the lithium ions to pass through.
During the charging phase, lithium ions move from the positive side of the battery to the negative side through the separator. While you discharge the battery, the ions move in the reverse direction.
This movement of lithium ions causes the electrical potential difference mentioned before. This electrical potential difference is called “voltage.” When you connect your electronics to a lithium battery, the electrons which are blocked by the separator are forced to pass through your device and power it.
Advantages of Lithium Battery
- Lightweight
- Ability to hold a charge
- Low self-discharge rates
- High energy density
- Very high current to applications
- Fast charging rates compared to lead acid batteries
Compared to the other high-quality rechargeable battery technologies (nickel-cadmium or nickel-metal-hydride), Li-ion batteries have a number of advantages. They have one of the highest energy densities of any battery technology today (100-265 Wh/kg or 250-670 Wh/L). In addition, Li-ion battery cells can deliver up to 3.6 Volts, 3 times higher than technologies such as Ni-Cd or Ni-MH. This means that they can deliver large amounts of current for high-power applications, which has Li-ion batteries are also comparatively low maintenance, and do not require scheduled cycling to maintain their battery life. Li-ion batteries have no memory effect, a detrimental process where repeated partial discharge/charge cycles can cause a battery to ‘remember’ a lower capacity. This is an advantage over both Ni-Cd and Ni-MH, which display this effect. Li-ion batteries also have low self-discharge rate of around 1.5-2% per month. They do not contain toxic cadmium, which makes them easier to dispose of than Ni-Cd batteries.
Due to these advantages, Li-ion batteries have displaced Ni-Cd batteries as the market leader in portable electronic devices (such as smartphones and laptops). Li-ion batteries are also used to power electrical systems for some aerospace applications, notable in the new and more environmentally friendly Boeing 787, where weight is a significant cost factor. From a clean energy perspective, much of the promise of Li-ion technology comes from their potential applications in battery-powered cars. Currently, the bestselling electric cars, the Nissan Leaf and the Tesla Model S, both use Li-ion batteries as their primary fuel source.
What is a lithium battery and how does it work?
A lithium-ion (Li-ion) battery is an advanced battery technology that uses lithium ions as a key component of its electrochemistry. During a discharge cycle, lithium atoms in the anode are ionized and separated from their electrons. The lithium ions move from the anode and pass through the electrolyte until they reach the cathode, where they recombine with their electrons and electrically neutralize. The lithium ions are small enough to be able to move through a micro-permeable separator between the anode and cathode. In part because of lithium’s small size (third only to hydrogen and helium), Li-ion batteries are capable of having a very high voltage and charge storage per unit mass and unit volume.
Li-ion batteries can use a number of different materials as electrodes. The most common combination is that of lithium cobalt oxide (cathode) and graphite (anode), which is most commonly found in portable electronic devices such as cellphones and laptops. Other cathode materials include lithium manganese oxide (used in hybrid electric and electric automobiles) and lithium iron phosphate. Li-ion batteries typically use ether (a class of organic compounds) as an electrolyte.
What Are The 6 Main Types Of Lithium Batteries?
Different types of lithium batteries rely on unique active materials and chemical reactions to store energy. Each type of lithium battery has its benefits and drawbacks, along with its best-suited applications.
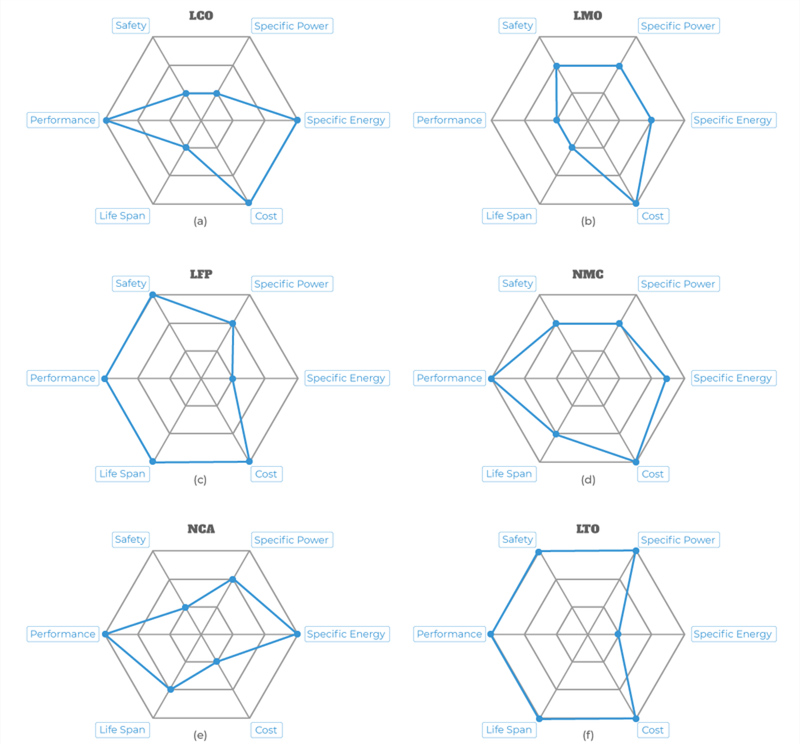
The different lithium battery types get their names from their active materials. For example, the first type we will look at is the lithium iron phosphate battery, also known as LiFePO4, based on the chemical symbols for the active materials. However, many people shorten the name further to simply LFP.
1. Lithium Iron Phosphate (LiFePO4) — LFP
Lithium iron phosphate (LFP) batteries use phosphate as the cathode material and a graphitic carbon electrode as the anode. LFP batteries have a long life cycle with good thermal stability and electrochemical performance.
What Are They Used For:
LFP battery cells have a nominal voltage of 3.2 volts, so connecting four of them in series results in a 12.8-volt battery. This makes LFP batteries the most common type of lithium battery for replacing lead-acid deep-cycle batteries.

Benefits:
There are quite a few benefits to lithium iron phosphate batteries that make them one of the most popular options for applications requiring a large amount of power. The primary benefits, however, are durability, a long life cycle, and safety.
LFP batteries typically have a lifecycle rating of 2,000 cycles or more. Unlike lead-acid batteries, depth of discharge has a minimal impact on the lifespan of LFP batteries. Most LFP manufacturers rate their batteries at 80% depth of discharge, and some even allow 100% discharging without damaging the battery.
The materials used in lithium iron phosphate batteries offer low resistance, making them inherently safe and highly stable. The thermal runaway threshold is about 518 degrees Fahrenheit, making LFP batteries one of the safest lithium battery options, even when fully charged.
Drawbacks:
There are a few drawbacks to LFP batteries. The first is that compared to other lithium battery types, they have a relatively low specific energy. Their performance can also suffer in low temperatures. Combining the low specific energy and reduced performance in cold temperatures means LFP batteries may not be a great fit in some high cranking applications.
2. Lithium Battery Cobalt Oxide (LiCoO2) — LCO
Lithium cobalt oxide (LCO) batteries have high specific energy but low specific power. This means that they do not perform well in high-load applications, but they can deliver power over a long period.
What Are They Used For:
LCO batteries were common in small portable electronics such as mobile phones, tablets, laptops, and cameras. However, they are losing popularity to other types of lithium batteries due to the high cost of cobalt and concerns around safety.

Benefits:
The key benefit to LCO batteries is their high specific energy. This allows them to deliver power over a relatively long period under low-load applications.
Drawbacks:
LCO batteries have some significant drawbacks resulting in them becoming less popular in recent years. First, LCO batteries suffer from a relatively short lifespan, usually between 500-1,000 cycles. Additionally, cobalt is fairly expensive. Expensive batteries that don’t last a long time are not cost-effective.
LCO batteries also have low thermal stability, which leads to safety concerns. Furthermore, their low specific power limits the ability of LCO batteries to perform in high-load applications.
3. Lithium Battery Manganese Oxide (LiMn2O4) — LMO
Lithium Manganese Oxide (LMO) batteries use lithium manganese oxide as the cathode material. This chemistry creates a three-dimensional structure that improves ion flow, lowers internal resistance, and increases current handling while improving thermal stability and safety.
What Are They Used For:
LMO batteries are commonly found in portable power tools, medical instruments, and some hybrid and electric vehicles.

Benefits:
LMO batteries charge quickly and offer high specific power. This means they can deliver higher current than LCO batteries, for example. They also offer better thermal stability than LCO batteries, meaning they can operate safely at higher temperatures.
One other benefit to LMO batteries is their flexibility. Tuning the internal chemistry allows LMO batteries to be optimized to handle high-load applications or long-life applications.
Drawbacks:
The main downside to LMO batteries is their short lifespan. Typically, LMO batteries will last 300-700 charge cycles, significantly fewer than other lithium battery types.
4. Lithium Battery Nickel Manganese Cobalt Oxide (LiNiMnCoO2) — NMC
Lithium nickel manganese cobalt oxide (NMC) batteries combine the benefits of the three main elements used in the cathode: nickel, manganese, and cobalt. Nickel on its own has high specific energy but is not stable. Manganese is exceptionally stable but has a low specific energy. Combining them yields a stable chemistry with a high specific energy.
What They Are Used For:
Similar to LMO batteries, NMC batteries are popular in power tools as well as electronic powertrains for e-bike, scooters, and some electric vehicles.

Benefits:
The benefits of NMC batteries include high energy density and a longer lifecycle at a lower cost than cobalt-based batteries. They also have higher thermal stability than LCO batteries, making them safer overall.
Drawbacks:
The major drawback to NMC batteries is that they have a slightly lower voltage than cobalt-based batteries.
5. Lithium Battery Nickel Cobalt Aluminium Oxide (LiNiCoAlO2) — NCA
Lithium nickel cobalt aluminum oxide (NCA) batteries offer high specific energy with decent specific power and a long lifecycle. This means they can deliver a relatively high amount of current for extended periods.
What They Are Used For:
The ability to perform in high-load applications with a long battery life makes NCA batteries popular in the electric vehicle market. Specifically, NCA is the battery of choice for Tesla.
Benefits:
The biggest benefits of NCA batteries are high energy and a decent lifespan.
Drawbacks:
With NCA technology, the batteries aren’t as safe as most other lithium technologies and are expensive in comparison.
6. Lithium Battery Titanate —LTO
All of the previous lithium battery types we have discussed are unique in the chemical makeup of the cathode material. Lithium titanate (LTO) batteries replace the graphite in the anode with lithium titanate and use LMO or NMC as the cathode chemistry.
The result is an extremely safe battery with a long lifespan that charges faster than any other lithium battery type.
What Are They Used For:
Many applications use LTO batteries. Electric vehicles and charging stations, uninterrupted power supplies, wind and solar energy storage, solar street lights, telecommunications systems, and aerospace and military equipment are just some of the use cases.

Benefits:
LTO batteries offer many benefits, including fast charging, an extremely wide operating temperature, a long lifespan, and superb safety because of their stability.
Drawbacks:
There are a couple of significant hurdles for LTO batteries to overcome. They offer low energy density, which means it stores a lower amount of energy relative to its weight when compared to some other lithium technologies. Additionally, they are very expensive.
Do All Types of Batteries Use Lithium Battery?
No, not all batteries use lithium. Lithium batteries are relatively new and are becoming increasingly popular in replacing existing battery technologies.
One of the long-time standards in batteries, especially in motor vehicles, is lead-acid deep-cycle batteries. Lithium has quickly gained ground in this market in recent years, but lead-acid is still the primary choice in gas-powered motor vehicles due to the low upfront cost.

Additionally, the most common types of off-the-shelf batteries found in stores are alkaline batteries. Most of the AA and AAA batteries in use today are alkaline batteries that use zinc and manganese dioxide for the chemical reaction to store energy.
Before rechargeable lithium batteries gained popularity, most rechargeable batteries were nickel-cadmium (NiCad). NiCad batteries use nickel oxide hydroxide and metallic cadmium as electrode materials. While not entirely obsolete yet, NiCad batteries are becoming less popular as lithium batteries take over the rechargeable battery market.
What’s The Most Common Type of Lithium Battery?
Lithium cobalt oxide (LCO) batteries are used in cell phones, laptops, tablets, digital cameras, and many other consumer-facing devices. It should be of no surprise then that they are the most common type of lithium battery.

Choose The Right Lithium Battery For Your Job
As you can see, there are many different types of lithium batteries. Each one has pros and cons and various specific applications they excel in. Your application, budget, safety tolerance, and power requirements will determine which lithium battery type is best for you.
LifePO4 Batteries Related Products:
Related Products Application:
LifePO4 Batteries Related Posts:
Tag in this article: #LiFePO4 Battery
Tips: more detail information, for deep cycle LiFePO4 battery。