Lithium Battery Knowledge
Lithium batteries are already closely related to our lives, and our food, clothing, housing and travel are inextricably linked to them. However, many people do not understand lithium batteries. This is not limited to ordinary consumers. Even the designers of various electrical appliances have a smattering of knowledge about lithium batteries. The following will share a series of articles on lithium battery knowledge. Through these articles, everyone will have a systematic understanding of lithium batteries.
The lithium-ion battery mentioned here refers specifically to the secondary lithium-ion battery that can be recharged repeatedly, rather than the primary battery that is thrown away when it is used up. Lithium-ion batteries are distributed in every corner of our lives. Their application areas include mobile phones, tablet computers, notebook computers, smart watches, mobile power supplies (power banks), emergency power supplies, razors, electric bicycles, electric vehicles, electric buses, tourist vehicles, drones, and other power tools.As the carrier of electrical energy and the source of power for many equipment, it can be said that without lithium-ion batteries, today’s material world will not be able to play around (unless we want to go back decades).So, what is a lithium battery?
This article does not popularize the basic principles and development history of batteries. If you are interested, please check on Baidu. There are many stories here.The basic theories in the fields of physics and chemistry were basically confused by the wave of people before Einstein. Batteries are directly related to these two fields. The theories related to batteries were almost studied before World War II, and there was no major innovation after World War II.As a kind of battery technology, the theoretical research on lithium battery has not made any breakthroughs in recent years. Most of the research has focused on materials, formulas, processes, etc., that is, how to improve the degree of industrialization and develop lithium-ion batteries with better performance (store more energy and last longer).
How to Choose a Carrier of Energy
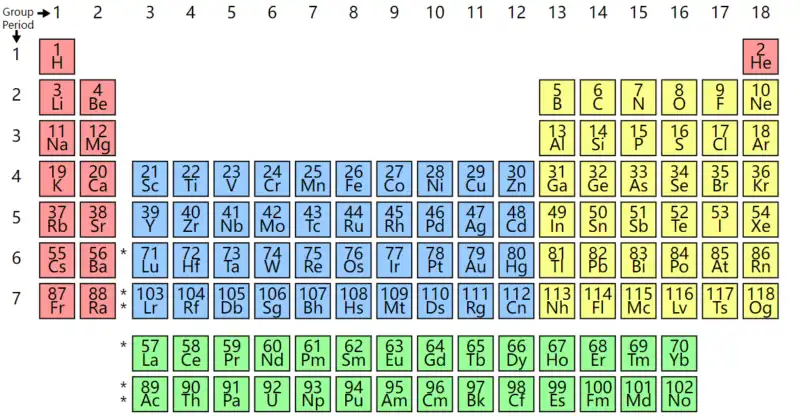
First of all, everyone will ask, why choose lithium as an energy carrier? Well, although we don’t want to review the knowledge of chemistry, we have to go to the periodic table to find the answer to this question. Fortunately, everyone always remembers the periodic table, right?!I really don’t remember, let’s just take a minute to take a look at the table below.
- peridot-table for battery
If you want to be a good energy carrier, you must store and carry more energy in the smallest possible size and weight.Therefore, the following basic conditions need to be met:
1) The relative mass of atoms is smaller
2) Strong electronic ability to gain and lose
3) The proportion of electronic transfer should be high
Based on these three basic principles, the elements above the periodic table are better than the elements below, and the elements on the left are better than the elements on the right.For preliminary screening, we can only find materials in the first and second cycles of the periodic table: hydrogen, helium, lithium, beryllium, boron, carbon, nitrogen, oxygen, fluorine, and neon.Excluding inert gases and oxidants, only hydrogen, lithium, beryllium, boron, and carbon are left.
Hydrogen is the best energy carrier in nature, so the research of hydrogen fuel cells has always been in the ascendant, representing a very promising direction in the field of batteries.Of course, if nuclear fission technology can make major breakthroughs in the next few decades and can be miniaturized or even miniaturized, then portable nuclear fuel batteries will have broad room for development.
The next step is lithium. The choice of lithium as a battery is based on the relatively optimal solution we can find among all the current elements of the earth (the reserves of beryllium are too small, and it is a rare metal among rare metals).The dispute over the technical route of hydrogen fuel cells and lithium-ion batteries is in full swing in the field of electric vehicles, probably because these two elements are the better energy carriers we can find at present.Of course, there are also many commercial interests and even political games involved. These are not the areas to be discussed in this article.
By the way, the energy sources that already exist in nature and are widely used by mankind, such as oil, natural gas, coal, etc., are also mainly composed of carbon, hydrogen, oxygen and other elements (in the first and second cycles of the periodic table).Therefore, whether it is a natural choice or a human “design”, it will eventually go the same way.
How Lithium-ion Batteries Work
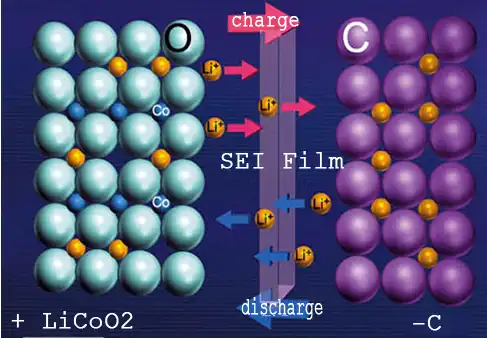
Let’s talk about the working mechanism of lithium battery. The redox reaction is not described here. Those with a bad chemical foundation, or those who have returned their chemical knowledge to the teacher, will feel dizzy when they see these professional things, so we should make a straightforward description.Borrow a picture here, this picture is easier for people to understand the principle of lithium battery.
- film for Lithium battery
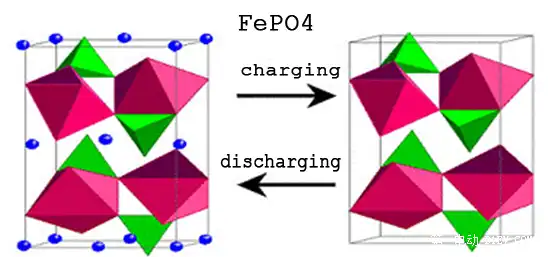
According to the habit of use, we distinguish the positive electrode (+) and the negative electrode (-) according to the voltage difference during charge and discharge. The anode and cathode are not mentioned here, which is time-consuming and laborious.In this picture, the cathode material of the battery is lithium cobalt oxide (LiCoO2) and the anode material is graphite (C).
When charging, under the influence of an external electric field, the lithium element in the molecule of the cathode material LiCoO2 is detached and becomes a positively charged lithium ion (Li+). Under the action of the electric field force, it moves from the positive electrode to the negative electrode, and reacts chemically with the carbon atoms of the negative electrode to generate LiC6, so the lithium ions that run out of the positive electrode are very “stably” embedded in the graphite layered structure of the negative electrode.The more lithium ions that run out of the positive electrode and transfer to the negative electrode, the more energy this battery can store.
When discharging, it is just the opposite. The internal electric field turns, and the lithium ion (Li+) detaches from the negative electrode. Following the direction of the electric field, it runs back to the positive electrode and becomes a lithium cobalt oxide molecule (LiCoO2) again.The more lithium ions that run out of the negative electrode and transfer to the positive electrode, the more energy this battery can release.
During each charge and discharge cycle, lithium ions (Li+) act as the carrier of electrical energy, moving back and forth from the positive electrode to the negative electrode to the positive electrode over and over again, chemically reacting with the positive and negative electrode materials, converting chemical energy and electrical energy to each other, and realizing the transfer of charge. This is the basic principle of “lithium-ion battery”.Since electrolytes, isolation membranes, etc. are all insulators of electrons, there is no movement of electrons back and forth between the positive and negative electrodes during this cycle, they only participate in the chemical reaction of the electrodes.
The Basic Composition of Lithium Battery
To achieve the above functions, lithium battery need to contain several basic materials inside: positive electrode active substance, negative electrode active substance, isolation film, electrolyte.Let’s make a brief discussion below, what are these materials for?
It is not difficult to understand the positive and negative electrodes. To achieve charge movement, a positive and negative electrode material with a potential difference is required. So what is an active substance?We know that batteries actually convert electrical energy and chemical energy to each other to achieve energy storage and release.To realize this process, it is necessary that the positive and negative materials are “easy” to participate in chemical reactions, to be active, to be easy to oxidize and reduce, so as to achieve energy conversion, so we need “active substances” to be the positive and negative electrodes of the battery.
As already mentioned above, lithium is our preferred material for batteries, so why not use lithium metal as the active substance for electrodes?Isn’t this the maximum energy density that can be achieved?
Let’s look at the picture above again. The three elements oxygen (O), cobalt (Co), and lithium (Li) constitute a very stable cathode material structure (the proportion and arrangement in the figure are for reference only), and the carbon atom arrangement of the anode graphite also has a very stable layered structure.Positive and negative electrode materials must not only be lively, but also have a very stable structure in order to achieve orderly and controllable chemical reactions.What is the unstable result?Think about the burning of gasoline and the explosion of bombs, and the violent release of energy. The process of chemical reaction is actually impossible to accurately control artificially, so the chemical energy becomes heat energy, and the energy is released at once, and it is irreversible.
- charging and discharging for lifepo4 battery
The lithium element in the metal form is too “lively”, and most naughty children are disobedient and like to destroy it.Early research on lithium batteries did focus on lithium metal or its alloy as the negative electrode, but because of outstanding safety issues, other better paths had to be found.In recent years, with people’s pursuit of energy density, this research direction has a trend of “resurrection with blood”, which we will talk about later.
In order to achieve chemical stability in the process of energy storage and release, that is, the safety and long life of the battery charge and discharge cycle, we need an electrode material that is lively when it needs to be lively and stable when it needs to be stable.After long-term research and exploration, people have found several metal oxides of lithium, such as lithium cobalt oxide, lithium titanate, lithium iron phosphate, lithium manganese oxide, nickel-cobalt-manganese ternary and other materials, as the active substance of the positive or negative electrode of the battery, to solve the above problems.As shown in the figure above, the peridot structure of lithium iron phosphate is also a very stable cathode material structure. The de-embedding of lithium ions during the charging and discharging process does not cause the lattice to collapse.Off topic, lithium metal batteries are indeed available, but compared with lithium-ion batteries, they are almost negligible. The development of technology will ultimately serve the market.
Of course, while solving the stability problem, it also brought serious “side effects”. That is, the proportion of lithium as an energy carrier was greatly reduced, and the energy density was reduced by more than an order of magnitude. Gains and losses, the natural way.
Graphite or other carbon materials are usually used as active substances for negative electrodes. They also follow the above principles. They require not only a good energy carrier, but also relatively stable, and relatively rich reserves, which are easy to manufacture on a large scale. Looking around, carbon is a relatively optimal solution.Of course, this is not the only solution. The research on anode materials is extensive, and it will be discussed later.
What do electrolytes do?In layman’s terms, it is the “water” in the swimming pool that allows lithium ions to swim around freely. Therefore, the ion conductivity should be high (the resistance to swimming is small), the electronic conductivity should be small (insulation), the chemical stability should be good (stability is overwhelming), the thermal stability should be good (all for safety), and the potential window should be wide.Based on these principles, after long-term engineering exploration, people have found electrolytes made of high-purity organic solvents, electrolyte lithium salts, and necessary additives. Electrolytes are formulated under certain conditions and in a certain proportion.Organic solvents include PC (propylene carbonate), EC (vinyl carbonate), DMC (dimethyl carbonate), DEC (diethyl carbonate), EMC (methyl ethyl carbonate) and other materials.Electrolyte lithium salts have LiPF6, LiBF4 and other materials.
The isolation film is added to prevent direct contact between the positive and negative electrode materials. We hope to make the battery as small as possible and store as much energy as possible, so the distance between the positive and negative electrodes is getting smaller and smaller, and short circuit has become a huge risk.In order to prevent the positive and negative electrode materials from shorting and causing a violent release of energy, it is necessary to use a material to “isolate” the positive and negative electrodes. This is the origin of the isolation film.The isolation film needs to have good ion permeability, mainly to open a channel for lithium ions to pass freely, and at the same time it is an insulator for electrons to achieve insulation between the positive and negative electrodes.At present, the main diaphragms on the market are single-layer PP, single-layer PE, double-layer PP/PE, three-layer PP/PE/PP composite film, etc.
The complete material composition of lithium battery
In addition to the four main materials mentioned above, in order to turn lithium-ion batteries from an “experimental product” in the laboratory into a product that can be used commercially, some other indispensable materials are also needed.
In addition to active substances, there are conductive agents and binders, as well as substrates and collectors used as current carriers (the positive electrode is usually aluminum foil).The binder should uniformly “fix” the lithium metal oxide as the active substance on the positive electrode substrate, and the conductive agent should enhance the conductivity of the active substance and the substrate to achieve a greater charge and discharge current. The collector is responsible for acting as a charge transfer bridge inside and outside the battery. The structure of the negative electrode is basically the same as that of the positive electrode. A binder is required to fix the active substance graphite, and copper foil is required as the substrate and the collector to act as the conductor of current.
However, because graphite itself has good electrical conductivity, the negative electrode generally does not add conductive agent material. In addition to the above materials, a complete lithium-ion battery also includes an insulating sheet, a cover plate, a pressure relief valve, a housing (aluminum, steel, composite film, etc.), and other auxiliary materials.
Production process of lithium battery
The production process of lithium-ion batteries is more complicated, and only some of the key processes are briefly described here.Depending on the assembly method of the pole piece, there are usually two process routes: winding and laminating.
The lamination process is to cut the positive and negative electrodes into small pieces and stack the isolation film to synthesize small cell monomers, and then stack the small cell monomers in parallel to form a large cell manufacturing process. The general process flow is as follows:
- process of lithium battery
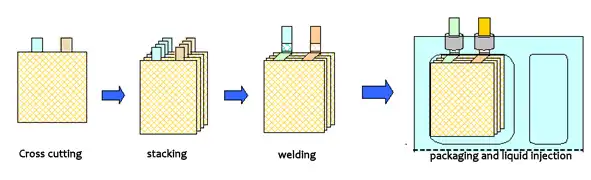
- process of lithium battery
The winding process is to fix the positive and negative electrode sheet, isolation film, positive and negative electrode ear, protective tape, termination tape and other materials on the equipment, and the equipment is unwound to complete the battery cell production.
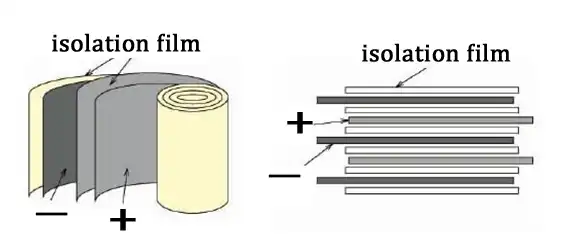
- process of lithium battery 3

- process of lithium battery 4
The common shapes of lithium-ion batteries are mainly cylindrical and square. Depending on the housing material, there are metal housings and soft-packed housings. Today, our sharing is over. We will give more articles about lithium batterirs. Stay tuned
Mos in Lithium Battery : Roles and Functions
Lithium batteries are a class of batteries that are made of lithium metal or lithium alloy as the positive/negative electrode material and use a non-aqueous electrolyte solution.In 1912, lithium metal batteries were first developed by Gilbert N. Lewis proposed and researched.In the 1970s, M. S. Whittingham proposed and began to study lithium-ion batteries.Because the chemical properties of lithium metal are very active, the processing, preservation and use of lithium metal have very high environmental requirements.With the development of science and technology, lithium batteries have become the mainstream.
Lithium batteries can be roughly divided into two categories: lithium metal batteries and lithium-ion batteries.Lithium-ion batteries do not contain lithium in the metallic state and can be charged.The fifth-generation product of rechargeable batteries, lithium metal batteries, was born in 1996, and its safety, specific capacity, self-discharge rate, and performance-to-price ratio are better than lithium-ion batteries.
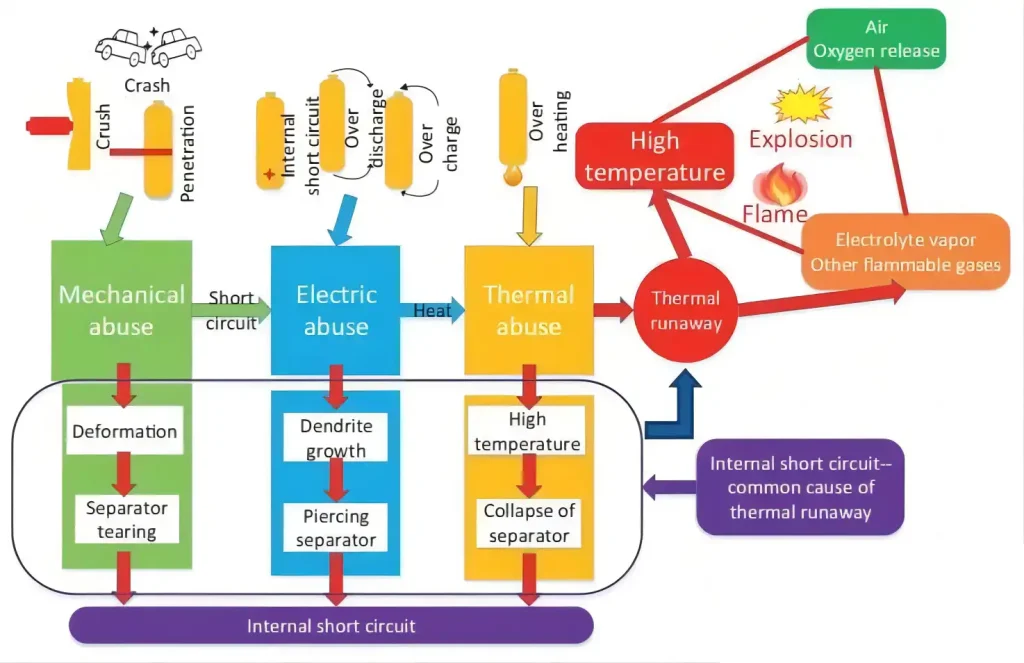
With the introduction of the new national standard and the support of national policies, the sales of lithium batteries have hit a boom, but the number of accidents caused by the safety of lithium batteries has also risen, and the consequences of many problems are shocking. Frequent lithium battery fires and explosions have once again sounded the alarm about the safety of lithium batteries.Therefore, lithium batteries have a safe voltage range for use. The highest and lowest voltages are generally called the charge-discharge termination voltage or cut-off voltage. When the actual operating voltage of the battery is lower than the discharge termination voltage for a long time or higher than the charge termination voltage for a long time, irreversible damage will occur inside the battery, seriously harming the battery, resulting in performance degradation, commonly known as battery attenuation, the performance of battery attenuation is the increase of the internal resistance of the battery, the capacity decreases, etc.Therefore, generally, there will be a small PCB board inside the lithium-ion battery, which is packaged with the battery. As shown in the figure below, the main function is to protect the battery.
Function of MOS in Lithium Battery
(1) Detect overcharging
(2) Detected discharge
(3) Detect over-current during charging
(4) Detect the over-current during discharge
(5) Detect over-current during short circuit
(6) Battery CELL longevity
(7) Restrictions of the external environment
(8) Over-current detection during charge and discharge and the rated current of the power MOSFET for charge and discharge control
After MOS is turned on, there is an on-resistance, so that the current will consume energy on this resistance. This part of the energy consumed is called conduction loss.Choosing a MOS tube with a small on-resistance will reduce the on-loss.The current on-resistance of low-power MOS tubes is generally around tens of milliohm, and there are also a few milliohm.
When MOS is turned on and off, it must not be done in an instant.The voltage across the MOS has a process of falling, and the current flowing through has a process of rising. During this period of time, the loss of the MOS tube is the product of voltage and current, which is called switching loss.Usually the switching loss is much greater than the conduction loss, and the faster the switching frequency, the greater the loss.
The product of voltage and current at the moment of conduction is very large, and the loss caused is also very large.Shortening the switching time can reduce the loss each time it is turned on; reducing the switching frequency can reduce the number of switches per unit time.Both of these methods can reduce the switching loss.
The working process of the MOS tube is very complicated, and there are many variables in it. In short, slow switching is not easy to cause Miller shock (introduction to Miller capacitance, Miller effect, etc., very detailed), but the switching loss will increase and the heat generation will be large; the switching speed is fast, and the loss will be reduced, but the Miller shock is very severe, which will increase the loss.The wiring of the drive circuit and the wiring of the main circuit are very demanding. In the end, it is to find a balance point. The general opening process does not exceed 1us.
In all, the lower the internal resistance of the protection plate, the better, and the lower the lower the less heat is generated.The current limiting thickness of the protection board is determined by the sampling resistance of the Kang copper wire, but the continuous current is determined by the mos, so it is very important to choose the appropriate mos model.
PTC in Lithium Battery
We know that as a large number of electric vehicles are put into use, the quality problem of poor battery consistency is gradually being exposed. Data from the State Administration of Market Supervision and Administration show that in 2018, there were at least 40 fire accidents involving new energy vehicles in our country, which is equivalent to an average of about 3 fire accidents per month. At the same time, new energy vehicles still have outstanding safety issues and high recall rates for three-electric vehicles. The safety situation of new energy vehicles deserves vigilance.
From the perspective of the chemical structure of the lithium battery itself, the fire and explosion of the lithium battery is caused by the thermal runaway of the lithium battery. The root cause is mainly the violent chemical exothermic reaction between the electrode and the electrolyte, caused by the unlimited diffusion of heat.
Studies have shown that in the thermal analysis of the battery system, the thermal stability of lithium iron phosphate is the best in terms of material. The safety of the battery first depends on the safety of its own materials. To increase the safety of batteries, high-energy batteries may need to include specific protection devices in the system design, such as cooling systems, explosion-proof systems, etc.
At the monomer level, in addition to conventional thermal safety design, it is more important to establish monomer self-excitation thermal protection. Let the monomer adjust its current output or power output according to the temperature it feels. If the battery can turn off the reaction, its heat production will be terminated.
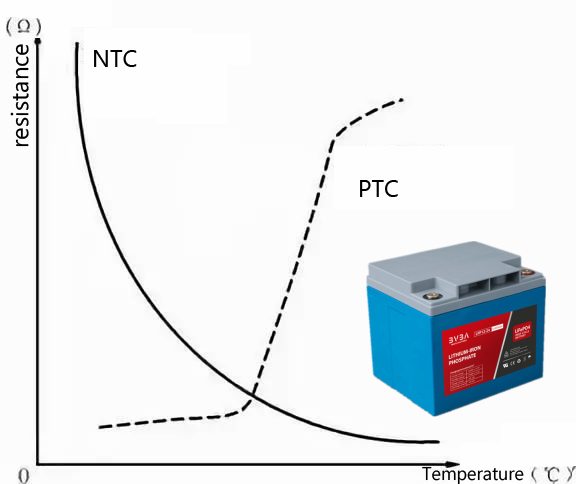
Among them, the PTC thermistor material that plays the role of temperature sensor monitoring has an important feature that when the temperature rises to a certain extent, the material will change from a good conductive state to an insulating state, which will be one of the important paths in monomer thermal protection technology.
The Causes of Lithium Battery Failure
The main failure methods of batteries during transportation and use include mechanical indiscriminate use (impact, collision, acupuncture, etc. ), electrical indiscriminate use (overcharge, over discharge, etc. ), thermal indiscriminate use (overheating), etc. These reasons will cause the destruction of the positive and negative electrode structure of the internal material of the battery, the thermal decomposition of the SEI film, the decomposition and exothermic of the electrolyte, and the heat shrinkage of the diaphragm, resulting in a short circuit inside the battery. When a violent chemical or electrochemical reaction occurs inside the battery, a large amount of heat accumulation will occur, causing serious thermal runaway. At present, the safety protection for lithium batteries can be mainly divided into: external protection, built-in self-excitation protection, and improved material stability. External protection mainly includes setting up a safety valve on the battery housing, external series PTC components, hot melt fuses and BTMS thermal management.
- lithium-battery-failure
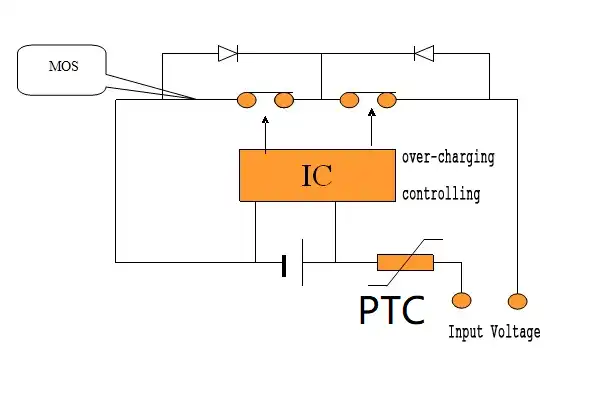
What is PTC in Lithium Battery?
PTC thermistor, that is, positive temperature coefficient thermistor, that is, PTC (positive temperature coefficient). It is a type of temperature sensor material whose resistance value increases with temperature. The application of PTC in lithium-ion batteries has been studied for many years, and it is currently widely used in some consumer batteries and power batteries. After installing a PTC thermistor element in a lithium-ion battery, when the battery heats up due to overcharge, the internal resistance of the battery increases rapidly, thereby limiting the current and realizing automatic protection of the battery.
- PTC in Lithium Battery
Externally protected PTC components are often connected in series on the battery housing. The working principle is that when the external current of the battery is overloaded, the temperature of the PTC element exceeds its Curie temperature, causing the resistance of the PTC element to increase rapidly by several orders of magnitude, thereby achieving the current limiting effect. Due to the slow heat transfer speed of the internal materials of the battery, the temperature difference between the inside and outside of the battery is often large. The PTC element connected in series on the housing cannot respond to the internal temperature in a timely manner, so it cannot effectively suppress the thermal runaway of the battery.
Specifically, the PTC thermistor element is installed in the lithium-ion battery, taking into account the pressure and temperature inside the battery. When the battery is heated up due to overcharge, the internal resistance of the battery increases rapidly, thereby limiting the current and reducing the voltage between the positive and negative electrodes to a safe voltage, realizing the automatic protection function of the battery.
The Basic Requirements for PTC Materials:
- -High conductivity at room temperature, does not affect the electro chemical properties of the battery within the normal operating temperature range;
- -Stable chemical and electro chemical properties, does not react with other components of the battery, and has good compatibility;
- -PTC thermal response speed is fast;
- -The resistance change temperature is appropriate (100~130℃), and the lift-to-resistance ratio is large.
Two Types of PTC used in Lithium Battery
PTC materials used in lithium batteries mainly include two types: one is based on a polymer polymer resin, which forms a composite with a conductive material. The corresponding substrates of this kind of polymer-based PTC are epoxy resin, polymethyl methacrylate, polyethylene-vinyl acetate, polyethylene, etc. ; Conductive agents are carbon black, nano-metal particles and various nano-fiber conductors. The working principle of the above-mentioned PTC material is: good contact between the conductive particles at room temperature, with high electrical conductivity; the expansion of the polymer matrix at high temperature increases the spacing of the conductive particles, the conductive network is destroyed, and the electronic conductivity of the complex drops sharply, showing PTC properties.
The other category is conductive polymers, including alkyl-substituted thiophene, alkylpyrrole, etc. The principle is that the doped conductive polymer has high electrical conductivity at room temperature, and the thermal movement of the polymer chain at high temperature causes the doped ions to be thermally impurity-free, resulting in a significant decrease in the conductivity of the polymer, showing PTC characteristics.
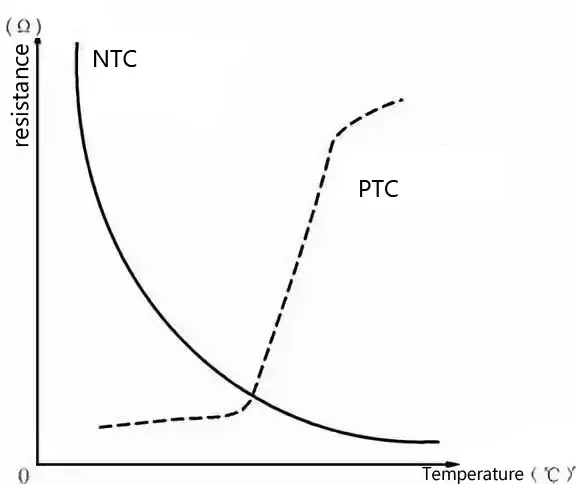
The Differences Between NTC and PTC
1.DIFFERENT RAW MATERIALS
PTC thermistors are resistors made of a mixture of barium titanate, titanium oxide, tantalum, silica and manganese. Within the temperature range, the resistance value increases as the temperature rises and decreases as the temperature decreases.
NTC thermistors are resistors sintered from two or more metal oxides such as manganese, copper, silicon, cobalt, and iron. Within the temperature range, the resistance value decreases as the temperature rises and increases as the temperature decreases.
- ptc-vs-ntc
2. DIFFERENT FUNCTIONS
PTC thermistors can also be used as heating elements. At the same time, they play the role of “switches”. They have three functions: sensitive components, heaters and switches, in addition to being used in industry for temperature measurement and control. They are called “thermal switches.”
NTC thermistors can be used for temperature measurement, temperature control, temperature compensation, etc., and can also be used for liquid level sensing.
It is undeniable that in the future and for some time to come, the safety of batteries will become more serious with the increase of the specific energy of new energy vehicle batteries. Summarizing the battery safety protection methods, it can be seen that positive temperature coefficient electrodes have great application prospects in the prevention of thermal runaway of lithium batteries. In the future battery preparation process streamline, the surface modification of the collector fluid (surface coating, surface roughening, corona treatment, etc. ) and the synthesis of surface PTC coating and other technologies are of great significance to obtain high magnification performance, high temperature resistance, and high cycle life lithium batteries. The research and development of suitable temperature-sensitive electrodes will continue to be a continuous hot spot in the development of lithium battery technology.
I believe that in the future, with the development of temperature sensor detection technology such as PTC thermistors, it will definitely help solve the old difficult problem of overheating protection of automotive lithium batteries. We will wait and see.
SM Bus in Lithium Battery
Smart battery, also known as Smart Battery System (SBS-Smart Battery System), is a branch and important part of modern power technology. A smart battery system consists of the following parts: system management bus (SMBus), smart battery charger and smart battery. SMBus is the abbreviation of System Magement Bus, which is translated as system management bus. It uses SMBus smart batteries. Its core concept is to transfer the charging control core from the charger to the battery, and the battery controls the charging process of the charger. This allows us to use only one universal charger to adopt a matching strategy for current and even future battery products to charge correctly.
This is only one aspect. Compared with traditional products, smart battery systems also have the following characteristics:
- High precision
The high-precision fuel gauge in the smart battery pack is different from the common voltage estimation method. Instead, it uses an advanced algorithm based on the electrochemical impedance of the battery. Even if the ambient temperature of the battery changes and the life of the battery ages, it can always accurately monitor the battery, even when the battery is not in the system.
- Intelligence
In the intelligent battery system, the host can obtain rich information about the battery, and the user can more accurately grasp the status of the battery (including the effective battery capacity, real-time voltage and current, historical cumulative charge and discharge times, remaining discharge time, predicted life, etc.), so as to achieve safer, more efficient and accurate battery management.In addition, it can also provide its production information, tell the system its model, manufacturer, etc.
- Security
Smart batteries have multiple redundant safety protections, which can realize battery safety protection even under the failure conditions of some devices. At the same time, the battery will permanently record battery failure information to facilitate the analysis of undesirable causes.
- Standardization
SBS has formed an international standard. The host designed with SBS architecture can minimize the non-repetitive engineering costs of hardware and software, and easily realize product upgrade iteration.
In fact, smart batteries have been widely used in many fields. Almost all notebook computers, as well as precision instruments, such as medical, precision measurement and aerospace technology, many devices use smart batteries to accurately control charge and discharge.
The interface of a standard smart battery is usually a 5PIN interface composed of +/C/D/T/-, where +/-is the positive and negative electrodes of the battery, which is connected to the load power supply port, and the remaining C and D are the clock lines and data lines for SMBus communication of the battery, which directly interact with the host microcontroller. Data, the T pin is the external temperature interface of the battery or the identification pin of the battery.Some specific types of tape recorders or power distributors can read the precise data of the battery in real time, and accordingly more accurately estimate the time when the equipment can continue to be used, so as to avoid accidental power outages.
What is SM Bus?
The system management bus (SMBus), defined by Intel in 1995, is a subset of I2C and defines stricter usage. One purpose of SMBus is to promote robustness and interoperability. Therefore, modern I2C systems incorporate some policies and rules from SMBus, and sometimes support both I2C and SMBus, and only need to use minimal re-configuration through commands or output pins.
As we all know, SMBus can be used in applications that require critical monitoring of parameters.Its most common applications are computer motherboards and embedded systems. It has additional monitoring specifications for temperature, power supply voltage, fan monitoring and/or control integrated chips.
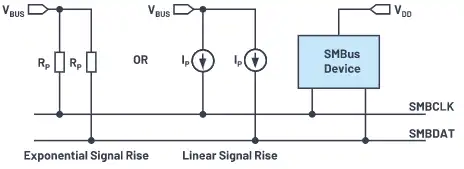
- SM-Bus-in-Circuit
SMBus is a 2-wire bus, similar to the I2C bus developed by Philips in the 1980s.The two main signals are clock (SMBCLK) and data (SMBDAT).I2C Primer and SMBus are compatible with each other, but there are obvious differences, such as:
- The SMBus logic level threshold is fixed and is not proportional to the power supply voltage of the device.Therefore, devices with different supply voltages can run on the same primer.For example, an SMBus may have multiple devices powered by 1.8 V, 3.3V, and 5 V power supplies.
- -They all run at the same speed of up to 100kHz, but I2C Primer is available in 400kHz and 2 MHz versions.
- -SMBus specifies the minimum clock speed and limits the amount of clock that can be extended in a transaction.Violation of the timeout limit will cause all SMBus devices to reset their I/O logic to allow the bus to restart.This design enhances the robustness of the bus.
- -The timeouts of the two are also different.I2C Primer does not have a timeout, while SMBus has a timeout-for the minimum clock speed of 10kHz, a timeout of 35 ms can be considered.
- -Packet error checking (PEC) was originally defined for SMBus.Add a grouping error code byte at the end of each transaction.
- -Some of the remaining differences involve transmission type, alarm line, pause line, shutdown or power-up.
Every time an SMBus device receives its own address, no matter what it is doing, it must answer (ACK). This is a clear requirement to ensure that the master device can accurately determine which devices on the bus are active.All SMBus transactions are executed through one of the specified SMBus protocols.SMBus also has an optional signal SMBALERT#, which can be used by the node device to quickly notify the master device or the system host. It has the information required by the master device, such as reporting a fault condition.
What is the relationship between I2C Primer, SMBus and PMBus?
SMBus was originally developed to assist in the battery management system, using I2C hardware, but a second level of software was added, which eventually allowed the device to be hot-swappable without restarting the system.PMBus extends SMBus and defines a set of device commands dedicated to managing power converters, exposing the measured voltage, current, temperature and other properties of the device.Generally speaking, I2C Primer, SMBus, and PMBus devices can share the bus without any major problems.
Advantages of I2C, SMB, and PMB
- Use only two lines
- -With ACK⁄NACK bit
- -Well-known agreement
- -Support multiple master devices and multiple nodes
- -The hardware is not as complicated as UART
- -Widely used method
Disadvantages
- Data transmission rate is slower than SPI
- -The size of the data frame is limited to 8 bits
- -The hardware required for implementation is more complicated than SPI
More Related Products Lithium Batteries Series
Lithium Battery Lithium-ion Battery