4 Types of Lead Acid Batteries
1. Wet (Flooded) Lead Acid Batteries
2. AGM Lead Acid Batteries
- Best for applications where short runtime is needed
- Eliminate the need for battery watering
- Eliminate risk of acid contact
- Short battery life
- Moderate cost lead acid battery
3. Gel Lead Acid Batteries
- Best for applications where short runtime is needed
- Eliminate the need for battery watering
- Typically higher cost than standard ($$$)
- Medium battery life
- Eliminate risk of acid contact
- Longer cycle life over standard AGM
4. TPPL (Thin Plate Pure Lead) Batteries:
Sealed lead acid batteries are widely used, but charging them can be a complex processas Tony Morgan explains:Charging Sealed Lead Acid (SLA) batteries does not seem a particularly difficult process, butthe hard part in charging an SLA battery is maximising the battery life. Simple constantcurrent / constant voltage chargers will do the job for a while, but the battery life expectancyquoted by the manufacturer will be greatly reduced by using non-intelligent chargers like this.Maximising the life of your SLA battery by using an intelligent charger is not only costeffective, it is also better for the environment.Before looking at the different charging techniques it is important to understand the batterychemistry and what happens during normal charge and discharge cycles.Typically the positive plates in an SLA battery are made from lead dioxide and the negativeplates from a sponge lead. The electrolyte is usually sulphuric acid mixed with a gelling agentand is largely absorbed and held by insulating separators between the plates, see Figure 1.
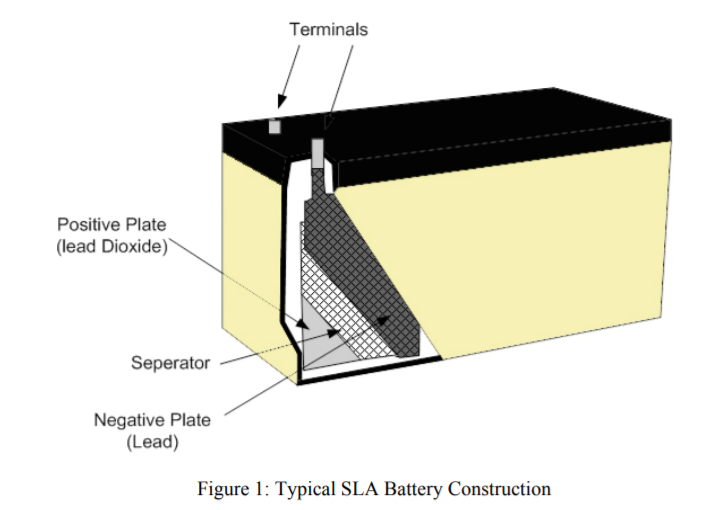
When an SLA battery is being discharged; the lead (Pb) on the negative plate and the leaddioxide (PbO2) on the positive plate are converted to lead sulphate (PbSO4). At the same timethe sulphuric acid (H2SO4) is converted to water (H2O).
In a normal charge, the chemical reaction is reversed. The lead sulphate and water are electrochemically converted to lead, lead dioxide and sulphuric acid. During a full charge cycle any gasses produced need to be re-combined in a so called ‘oxygen cycle’. Oxygen is generated atthe positive plates during the latter stages of the charge cycle, this reacts with and partiallydischarges in the sponge lead of the negative plates. As charging continues, the oxygen produced also re-combines with the hydrogen being produced on the negative plate formingwater. With correct and accurate cell voltage control all gasses produced during the charge Guide to charging Sealed Lead Acid batteriescycle will be re-combined completely into the negative plates and returned to water in the electrolyte.
If an SLA battery is over-charged, the excess cell voltage will result in the conversion ofelectrolyte into large amounts of hydrogen and oxygen gasses which cannot be recombined bythe normal processes. A pressure-release valve will open and vent the excess gas, resulting inthe loss of electrolyte and a loss of capacity.
If the battery is undercharged; the low cell voltage will cause the charge current to diminish to zero well before full capacity is reached. This will allow some of the lead sulphate produced during discharge to remain on the plates, where it will crystallise, which also causes apermanent loss of capacity.
It is also important to remember that SLA batteries have a self discharge rate ofapproximately 5% per month. This is less than most other forms of rechargeable batteries, buthas to be considered. Manufacturers recommend recharging when the battery reaches about70% of its capacity (approximately 2.1 volts per cell). They use this to calculate the maximumlife of the battery, but this is very difficult to implement in a real world application.
So let us look at different charging techniques: –
Constant Voltage Charging:
this method is the most commonly used for SLA batteries asthe individual cells tend to share the voltage and equalize the charge between them. It isimportant to limit the initial charging current to prevent damage to the battery. However, witha single fixed voltage, it is impossible to properly balance the requirements of a fast chargecycle against the danger of overcharge.
Constant Current Charging:
this method can be used for a single 2V cell but is notrecommended for charging a number of series connected cells, a battery, at the same time.This is because some cells will reach full charge before others and it is very difficult todetermine when the battery has reached a fully charged state. If the charge is continued at thesame rate, for any extended period of time, severe overcharge may occur to some cells,resulting in damage to the battery.
Taper Current Charging:
this method is not really recommended for charging SLA batteriesas it can often shorten battery service life due to poor control of the final fully chargedvoltage. However, because of the simplicity of the circuit and subsequent low cost, tapercurrent charging is often used to charge a number of series connected batteries that are subjectto cyclic use. When using this method it is recommended that the charging time is eitherlimited or that a charging cut-off circuit is incorporated to prevent overcharge.
Two Stage Constant Voltage Charging:
this method is a recommended for charging SLAbatteries in a short period of time and then maintaining them in a fully charged float (orstandby) condition.
Each of the above has its advantages and disadvantages, but using a simple charger designmay not be cost effective in the long term. Checking battery condition and replacing batteries with lost capacity is very costly and environmentally unfriendly. So designing a charger to maximise the life of the SLA battery is very important. Guide to charging Sealed Lead Acid batteries Another important factor that has to be considered when charging an SLA battery istemperature. As the temperature rises, electrochemical activity in a battery increases, so thecharging voltage should be reduced to prevent overcharge. Conversely as temperature falls,the change voltage should be increased to avoid undercharge.
Using a combination of the constant current charging and two stage constant voltage chargingtechniques and also by monitoring the battery terminal voltage and temperature a multi-stagecharge profile can be implemented to reduce stress on the battery while giving the shortest possible charge time.
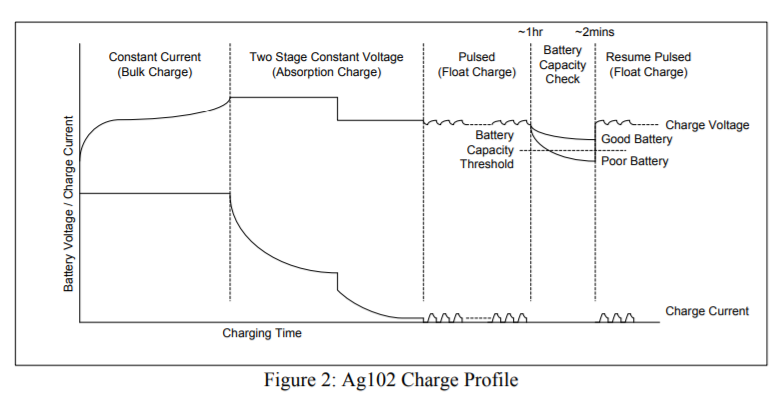
Figure 2 shows the multi-stage charge profile used by the Silvertel Ag102 module, for a 6-cellbattery that will achieve this. The upper trace shows the charge voltage and the lower traceshows the charge current.Charge CurrentCharge Voltage Battery Voltage / Charge Current Charging Time Constant Current (Bulk Charge) Two Stage Constant Voltage (Absorption Charge) Pulsed (Float Charge) Battery Capacity Check Resume Pulsed (Float Charge) Good Battery Poor Battery BatteryCapacity Threshold~1hr ~2mins
Over Charging
Over charging SLA batteries shortens their lifespan and can be dangerous. If you are not using a CC/CV charger, and the battery is left on the charger past the full charge, the battery can create excess gas and will diminish the capacity of the battery. The sulfuric acid will break down and form Hydrogen and Oxygen gases. In a sealed lead acid battery, this can result in the buildup of pressure and temperature. There is a safety valve that will vent the gas, but often some of the electrolyte solution is ejected as well, which reduces the capacity of the battery. The lost capacity of an overcharged SLA can’t be recaptured.
Because all SLA batteries can produce this gas, always charge them in a well ventilated area. Also keep in mind that a discharged battery will freeze faster than a charged battery due to the higher water content, so make sure the battery isn’t stored or charged in a cold setting, and always store batteries fully charged.
Sulfation
As a battery discharges, lead sulfate is produced, and it is reconverted to lead and lead dioxide as the battery recharges. But if the battery is stored without a full charge, or if the battery is never fully recharged, that lead sulfate may harden and then resist being converted back to lead dioxide and pure lead. The battery loses capacity as a result, and the lost capacity can’t be reversed. To avoid sulfation, make sure the battery is fully charged on most cycles.
Deep Discharge
SLA batteries do not tolerate repeated deep discharges well. If the battery does become completely discharged, all of the materials inside are converted to water and lead sulfate, and it might be difficult to reverse the chemical reaction to produce a charge. Some batteries designed to deal with deep discharges have an outsized plate that allows a recharge even when fully discharged. When possible, you should re-charge the battery after each use to avoid deep discharge.
Optimizing Battery Life
You can extend the life of your sealed lead acid battery if you are careful about charging it. More batteries are damaged by bad charging techniques than from all other causes. Check the manufacturer’s recommendation for charging and use the proper charger for the battery. Most chargers will only properly charge one type of SLA battery and shouldn’t be used for a battery with a different chemistry.
Using a CCCV charger, which uses a microprocessor to determine temperature, voltage and current SOC will reduce the risk of overcharging or under charging, and extend the battery’s useful lifespan
Tips for Charging Lead Acid Batteries
- Monitor the water levels: Do not let the water level fall below plates.
- Use the correct water type: When topping off your battery’s fill well, always use distilled or de-ionized water. Tap water typically contains minerals that can damage lead acid batteries and/or impact performance and lifespan.
- Start the day fully charged: Lead acid batteries should be charged every day after 15 minutes or more of use. Before using the following day, the machine must be plugged in and charged until the charger indicates the batteries are FULLY charged. Failure to allow the batteries to fully charge before the next use will diminish the life of the batteries.
- One full charge per day: Do not fully charge lead acid batteries more than once per 24-hour period to maximize your battery’s life. Opportunity charging, which means plugging in the machine for a short period of time without fully charging, can negatively impact the life of the batteries.
- Fully charge batteries before storing: Lead acid batteries should never be stored in a discharged state. Some of today’s machines place parasitic loads on the batteries. Even when the machine’s key is in the “OFF” position, there are electrical components drawing upon the battery’s energy.
- Check fluid levels: The flooded (wet) lead-acid batteries require routine watering. Check the battery electrolyte level weekly. The electrolyte level should be slightly above the battery plates as shown before charging. Add distilled water if low. Do not overfill. The electrolyte will expand and may overflow when charging. After charging, distilled water can be added up to about 3 mm (0.12 in) below the sight tubes.
- Use the correct charger: The battery charger is set to charge the battery type supplied with your machine. If you choose to change to a different battery type or capacity, the charger’s charging profile must be changed to prevent battery damage.
- Seek out new charger technology: Older lead acid battery chargers require careful monitoring to avoid “over-charging.” But new charger technology allows the batteries and charger to be plugged in over a weekend or longer. The charger will shut off once the full charge on batteries is reached. Some newer chargers can monitor the batteries and turn them back on as the batteries require a charge.
- Ideal charging conditions: Charge the batteries in a well-ventilated area of no more than 80 degrees Fahrenheit to prevent possible gas buildup. Never store or charge batteries in a place that is exposed to freezing temperatures, direct sunlight or heat, or other temperature extremes.
- Follow your operator manual: All of these battery charging best practices are fairly universal for all types of lead acid batteries. But, of course, be sure to read the operator manual for your Tennant cleaning machine for the specific charging protocols.
Simple Guidelines for Charging Lead Acid Batteries
- Charge in a well-ventilated area. Hydrogen gas generated during charging is explosive.
- Choose the appropriate charge program for flooded, gel and AGM batteries. Check manufacturer’s specifications on recommended voltage thresholds.
- Recharge lead acid batteries after each use to prevent sulfation. Do not store on low charge.
- The plates of flooded batteries must always be fully submerged in electrolyte. Fill the battery with distilled or de-ionized water to cover the plates if low. Never add electrolyte.
- Fill water level to designated level after charging. Overfilling when the battery is on low charge can cause acid spillage during charging.
- The formation of gas bubbles in a flooded lead acid indicates that the battery is reaching full state-of-charge. (Hydrogen appears on negative plate and oxygen on positive plate).
- Lower the float charge voltage if the ambient temperature is higher than 29°C (85°F)..
- Do not allow a lead acid to freeze. An empty battery freezes sooner than one that is fully charged. Never charge a frozen battery.
- Avoid charging at temperatures above 49°C (120°F).
Tag in this article: #Battery
Tips: more detail information, for acid battery。